If you’re looking for a quick answer:
👉 A mole (mol) is a unit used to count particles in chemistry, and 1 mole equals 6.022 × 10²³ particles.
This number is known as Avogadro’s number, and it’s the foundation for all mole conversions.
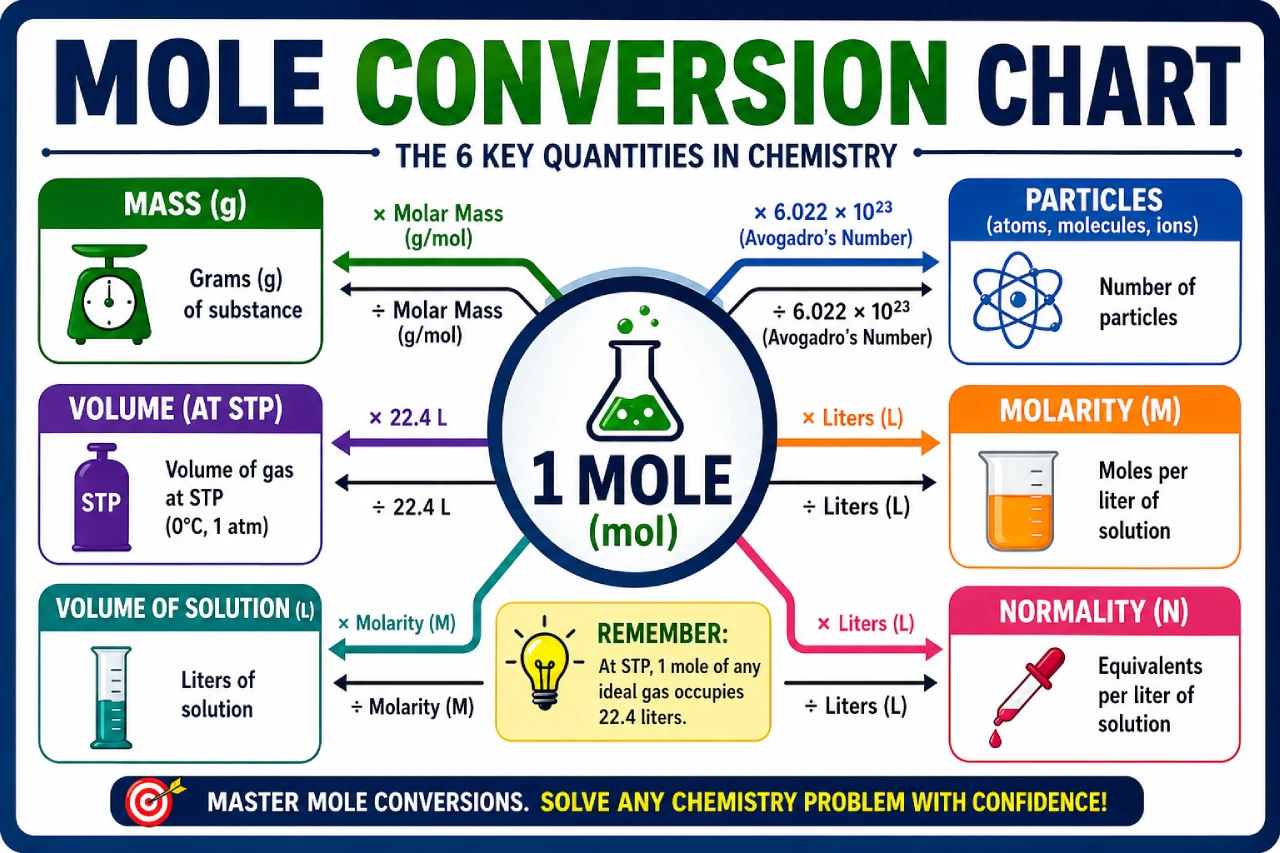
A mole conversion chart helps you quickly convert between:
- Moles and grams
- Moles and particles (atoms, molecules)
- Moles and volume (for gases)
In this guide, we’ll walk through a complete mole conversion chart, explain how to use it, and show step-by-step examples so you can confidently solve chemistry problems.
What Is a Mole in Chemistry?
A mole is a counting unit—just like a dozen means 12.
But instead of 12, a mole represents:
👉 6.022 × 10²³ particles
These particles could be:
- Atoms
- Molecules
- Ions
Why Use Moles?
Because atoms and molecules are incredibly small.
Instead of counting each one individually, chemists use moles to simplify calculations.
The Core Mole Relationships
Before looking at the chart, you need to understand the three main relationships.
1. Moles to Particles
👉 1 mole = 6.022 × 10²³ particles
2. Moles to Grams
👉 Moles = Mass (grams) ÷ Molar Mass
3. Moles to Volume (Gases)
👉 1 mole of gas = 22.4 liters (at STP)
These three relationships form the foundation of all mole conversions.
Mole Conversion Chart
Mole Conversion Chart & Converter
Convert between moles, particles, grams, liters of gas, and molarity using simple chemistry formulas.
Mole Conversion Chart
| Conversion | Formula |
|---|---|
| Moles to Particles | Moles × 6.022 × 10²³ |
| Particles to Moles | Particles ÷ 6.022 × 10²³ |
| Moles to Grams | Moles × Molar Mass |
| Grams to Moles | Grams ÷ Molar Mass |
| Moles to Liters at STP | Moles × 22.4 |
| Liters to Moles at STP | Liters ÷ 22.4 |
| Molarity | Moles ÷ Liters of Solution |
Here’s a simplified mole conversion chart you can use.
Basic Mole Conversion Chart
| From | To | Formula |
|---|---|---|
| Moles | Particles | Moles × 6.022 × 10²³ |
| Particles | Moles | Particles ÷ 6.022 × 10²³ |
| Moles | Grams | Moles × Molar Mass |
| Grams | Moles | Grams ÷ Molar Mass |
| Moles | Liters (gas) | Moles × 22.4 |
| Liters | Moles | Liters ÷ 22.4 |
This chart is your quick reference for solving most chemistry problems.
Understanding Molar Mass
To convert between moles and grams, you need molar mass.
What Is Molar Mass?
Molar mass is the mass of 1 mole of a substance, measured in grams per mole (g/mol).
Example
- Oxygen (O₂) = 32 g/mol
- Water (H₂O) = 18 g/mol
You can find molar mass using the periodic table.
Step-by-Step Examples
Let’s apply the chart in real problems.
Example 1: Moles to Particles
Convert 2 moles of carbon atoms into particles.
Step 1: Use the formula
Particles = Moles × Avogadro’s number
Step 2: Calculate
2 × (6.022 × 10²³) = 1.204 × 10²⁴ atoms
👉 Final Answer: 1.204 × 10²⁴ atoms
Example 2: Grams to Moles
Convert 36 grams of water (H₂O) into moles.
Step 1: Find molar mass
H₂O = 18 g/mol
Step 2: Use formula
Moles = Grams ÷ Molar Mass
Step 3: Calculate
36 ÷ 18 = 2 moles
👉 Final Answer: 2 moles
Example 3: Moles to Grams
Convert 3 moles of CO₂ into grams.
Step 1: Find molar mass
CO₂ = 44 g/mol
Step 2: Multiply
3 × 44 = 132 grams
👉 Final Answer: 132 grams
Example 4: Moles to Volume
Convert 1 mole of gas to volume at STP.
👉 1 mole = 22.4 liters
👉 Final Answer: 22.4 L
Why the Mole Conversion Chart Matters
This chart is essential for:
1. Chemical Reactions
Balancing equations requires mole relationships.
2. Lab Work
You need precise conversions for experiments.
3. Stoichiometry
Moles are the backbone of reaction calculations.
4. Real-World Applications
Used in:
- Pharmaceuticals
- Food science
- Environmental studies
Common Mistakes to Avoid
1. Forgetting Units
Always include units like g/mol or liters.
2. Using Wrong Molar Mass
Double-check calculations from the periodic table.
3. Mixing Up Formulas
Remember:
- Multiply when converting from moles
- Divide when converting to moles
Tips for Using the Mole Conversion Chart
Here are some practical tips.
Tip 1: Identify What You’re Given
Start with:
- Grams?
- Moles?
- Particles?
Tip 2: Identify What You Need
Find the target unit.
Tip 3: Use the Chart as a Map
Follow the correct formula step by step.
Tip 4: Cancel Units
Dimensional analysis helps avoid mistakes.
Visualizing the Mole Concept
Think of a mole like a “super-sized dozen.”
- 1 dozen = 12 items
- 1 mole = 6.022 × 10²³ items
This makes it easier to understand.
Advanced Mole Conversions
Sometimes conversions require multiple steps.
Example
Convert grams → moles → particles.
Steps
- Convert grams to moles
- Convert moles to particles
This is called multi-step conversion.
Frequently Asked Questions (FAQs)
What is a mole in chemistry?
A mole is a unit representing 6.022 × 10²³ particles.
What is Avogadro’s number?
👉 6.022 × 10²³
How do you convert moles to grams?
Multiply by molar mass.
How do you convert grams to moles?
Divide by molar mass.
What is molar mass?
The mass of 1 mole of a substance.
What is STP?
Standard Temperature and Pressure.
How many liters are in 1 mole of gas?
👉 22.4 liters (at STP)
Why is the mole important?
It simplifies counting atoms and molecules.
Can calculators solve mole problems?
Yes, but you must use the correct formulas.
Is mole used outside chemistry?
Mostly in scientific and industrial fields.
Final Thoughts
A mole conversion chart is one of the most powerful tools in chemistry.
Once you understand the three key relationships:
- Moles ↔ Particles
- Moles ↔ Grams
- Moles ↔ Volume
Everything else becomes easier.
The key takeaway is simple:
👉 Use the right formula based on what you’re converting
With practice, you’ll move through mole conversions quickly and confidently.
Now here’s a quick question for you:
Next time you see a chemistry problem, will you reach for a calculator—or let the mole conversion chart guide you step by step?
Leave a Reply